|
|
|
Int'l Pharma & Device Compliance Congress XIII Features Keynote by INSEAD Ethics Professor N. Craig Smith
|
- Sponsored by ETHICS, the International Society of Healthcare Compliance Professionals
- Media Partners: Policy & Medicine Compliance Update and Rx Data News
- April 8 - 10, 2019
- Onsite at the Grand Hyatt Athens Athens, Greece
- www.InternationalPharmaCongress.com
PRESS RELEASE
Phone: 800-503-8171
Email: registration@hcconferences.com
Website: www.InternationalPharmaCongress.com |
|
|
FEATURING WELCOME BY
|

Dimitra Lempesi, LLM, MBA
Legal Director, SFEE (Hellenic Association of Pharmaceutical Companies), Athens, Greece
|
|
|
|
AND ETHICS KEYNOTE BY
|

Professor N. Craig Smith
Chaired Professor of Ethics and Social Responsibility, INSEAD; Academic Director, CSR & Ethics Research Group, INSEAD Social Innovation Centre, Fontainebleau, France
|
|
|
|
ATHENS, GREECE -- PHARMA UPDATE NEWS SERVICE™ -- MARCH 28, 2019: The Thirteenth International Pharmaceutical and Medical Device Compliance Congress, www.InternationalPharmaCongress.com, will be held April 8 - 10, 2019 at the Grand Hyatt Athens in Athens, Greece.
|
|
KEYNOTE SPEAKERS
|

Julie Bonhomme
EFPIA Legal & Compliance Director, European Federation of Pharmaceutical Industries and Associations (EFPIA), Brussels, Belgium |

Rachel Cooper, LLB, MSc
Director, Pharmaceuticals & Healthcare Programme, Transparency International UK, Former Head, Government Relations, World Vision UK, Former Vice Consul, British Embassy, Copenhagen, Deputy Head of Mission, British Embassy, Bratislava, Deputy High Commissioner, Freetown, London, UK |

Holger Diener
Managing Director, Association of Voluntary Self-Regulation for the Pharmaceutical Industry ("FSA"), Member, Ethics and Compliance Committee and Vice Chair, Code Committee, EFPIA, Member, IFPMA Code Compliance Network, Berlin, Germany
|

Robert I. Dodge, JD
Assistant Director, FCPA Unit, US Securities & Exchange Commission, Former Assistant U.S. Attorney, US Attorney's Office, Western District of Michigan, Former Assistant Section Chief, Environmental Defense Section, US Department of Justice, Washington, DC, USA |

Emmanuel Farhat
Agence Fran�aise Anticorruption (AFA), France, Paris, France |

Abhiroop Gandhi
Trust and Compliance Officer, Verily Life Sciences (an Alphabet company), Former Vice President, Corporate Compliance, Mallinckrodt, San Francisco, CA, USA
|

Dimitra Lempesi, LLM, MBA
Legal Director, SFEE (Hellenic Association of Pharmaceutical Companies), Athens, Greece |

Sofie Melis
Head of Ethics and Compliance, International Federation of Pharmaceutical Manufacturers and Associations (IFPMA), Geneva, Switzerland |

Arthur Muratyan, Esq.
Secretary General, International Society of HealthCare Ethics and Compliance Professionals (ETHICS), Chair, MedTech Compliance Network, Former VP-Head of Legal Corporate, and Global Compliance Officer, Sanofi, Paris, France
|

Guido Palazzo, PhD
Professor of Business Ethics, Department of Strategy, Globalization and Society, University of Lausanne, Lausanne, Switzerland |

Piergiorgio Pepe, MA EU Law
President, Quantum Ethics, Ethics and Compliance Lecturer, SciencesPo, Former Compliance Director Western Europe & Canada, AbbVie, Former Director, Compliance & Ethics EMEA, Bristol-Myers Squibb, Paris, France |

Rami A. Rajab
Chairman, Mecomed Medical Technology, Association Middle East & Africa (MECOMED), Chairman, LIFE (LivaNova International Fellowship), International Corporate Social Responsibility, Former VP Market Access, Emerging Markets, LivaNova, Maidenhead, UK
|

Harout Semerjian, MBA
Executive Vice-President & Chief Commercial Officer, Ipsen, Former Senior Vice President and Global Launch Lead, Ribociclib, Novartis Oncology, New York, NY, USA |

Heather Simmonds
Director, Prescription Medicines Code of Practice Authority, London, United Kingdom
|

Professor N. Craig Smith
Chaired Professor of Ethics and Social Responsibility, INSEAD; Academic Director, CSR & Ethics Research Group, INSEAD Social Innovation Centre, Fontainebleau, France |

Christopher L. White, JD
Chief Operating Officer & General Counsel, Advanced Medical Technology Association (AdvaMed), Washington, DC, USA
|
|
|
|
FEATURING PRECONFERENCE SESSIONS
|
- Precon I: A Workshop on Practical Case Studies & Exercises in Ethics & Compliance
- Precon II: Advanced Investigations
|
|
PLENARY SESSIONS
|
- International Pharma Congress Vision and Overview
- Keynote Address: Pharma Executive
- Compliance Issues Raised by Pharma & Device New Business Models
- Global Compliance Codes Update
- Annual Chief Compliance Officer Roundtable
- Keynote Address: Transparency International
- Annual Anticorruption Roundtable
- Update on the new French Sapin II Law
- Annual FCPA Update Roundtable
- Multi-jurisdictional Anticorruption Investigations & Prosecutions
- Ethics and Compliance Keynote Address
- The Emerging Role of Technology Companies in Life Science
- IFPMA Global Compliance Updates
- Transforming Compliance into a Profession
|
|
|
|
AND CONCURRENT MINI SUMMITS
|
- Annual Medical Device Ethics & Compliance Roundtable
- Digitalization of Compliance
- Data Privacy, Consent Management and GDPR Update
- Ethics & Compliance Issues in Patient Relationships
- Annual Central and Eastern Europe (CEE) Compliance Update
- Annual Middle East Africa (MEA) Compliance Update
- EU & US Whistleblower Updates
- Using Risk Assessment to Design Compliance Programs
- Analytics with Reference to Transparency & Monitoring
- Improving the Future of Compliant HCP Engagements
- Reducing FCPA Exposure through Better Use of Data
- Compliance Issues in Manufacturer Relationships with Foundations
- Behavioral Science to Enhance Effectiveness of Compliance Programs
- Third Party Due Diligence and Management
- Structuring an Efficient Digital Compliance Framework
- Auditing and Monitoring Workgroup
- R&D and Clinical Trials Compliance Workgroup
- Working with in-company Business Partners Workgroup
|
|
|
FEATURED FACULTY
|

Pablo Rojas Abad, LLM
Manager, Legal and Compliance, MedTech Europe, Brussels, Belgium |

David Andersen
Partner, Pharmaceuticals and Life Sciences, PwC, London, UK |

Michael Bartke, PhD
Director Ethics & Compliance, Alexion, Former Director Compliance Management, Daiichi Sankyo Europe GmbH, Strategic Committee, ETHICS, Munich, Germany
|

Ann Beasley, JD
Director, Life Sciences Regulatory & Compliance, Navigant; Former Senior VP, CCO, Biogen, Boston, MA, USA |

Thomas W. Beimers, JD
Partner, Hogan Lovells; Former Senior Counsel for Administrative and Civil Remedies, Office of the Inspector General, Department of Health and Human Services; Minneapolis, MN, USA |

Maike Bermüller
Senior Legal Counsel and Global Healthcare Compliance Officer, Boehringer Ingelheim, Frankfurt Am Main, Germany
|

Chris van Bronckhorst
Global Compliance Director, IQVIA, The Hague, Netherlands |

Keith Burn
Global Investigations Director, Ipsen, Former Associate Investigator, Parliamentary and Health Service Ombudsman, NHS, Former Detective Constable, London Metropolitan Police, Amersham, Buckinghamshire, UK |

Antonio Cavallaro
Compilance & Data Protection Officer, Merck Serono SpA -- Italy; Former Head of Compliance, Takeda Italy, Rome, Italy
|

Matthew Coombs
Director, Account & Services Offering Management, Commercial Compliance, IQVIA, Former Strategic Business Director, Qforma, London, UK |

Michaeline Daboul
CEO & President, MMIS, Inc., New York, New York |

Peter Dieners, Esq.
Partner and Head, Global Healthcare and Life Sciences Group, Clifford Chance, Co-chair, Legal Affairs Focus Group (LA FG), EUCOMED, Co-chair, Compliance Network (CN), EUCOMED, Düsseldorf, Germany
|

Julien Durand, MBA, PhD
Executive Director Compliance & Ethics, Amgen, Former Chief Privacy Officer and Global Technology Compliance Head, AstraZeneca, Zürich, Switzerland |

Sue Egan
Director and Principal Consultant, Sue Egan Associates, Former Vice President Compliance, AstraZeneca, Great Missenden, Buckinghamshire, UK |

Karen Eryou
Head, Ethics & Compliance Programs, UCB Pharma, Former Co-chair, Asia Pacific Pharma Compliance Congress, Brussels, Belgium
|

George Fife
Partner, Fraud Investigation & Dispute Services, EY, Former Executive Director, Compliance & Ethics, Bristol-Myers Squibb, Paris, France |

Monique Fonfara-Weinberg
Compliance Manager, Boehringer Ingelheim Corporate Center GmbH, Ingelheim am Rhein, Germany |

Carlo Geninazza
Senior Manager, CAWECO GmbH, Munich, Germany
|

Gary F. Giampetruzzi, Esq.
Partner, Paul Hastings, Former Vice President and Assistant General Counsel, Head of Government Investigations, Pfizer Inc., New York, NY, USA |

Riccardo Giannetti
Scheme Manager, Inveo, Roma, Italy |

Betania Glorio
Global Compliance Officer Healthcare, Merck KGaA, Former Senior Director, Legal and Compliance EMEA, Horizon, Former Senior Counsel, Regional Compliance Director, EUCAN, Pfizer, Darmstadt, Germany
|

Cecile Gousset
Head of Compliance Risk Assessment, Policies & Education, Global Ethics & Business Integrity, Sanofi, Paris, France |

Tom Gregory, MBA
Partner, Forensic & Integrity Services, EY, Atlanta, USA |

Ulf H. Grundmann
Partner, FDA and Life Sciences, King & Spalding LLP, Frankfurt am Main, Germany
|

Thomas K. Hauser, MA HSG, MSJ
Director, Life Sciences Governance, Risk Management and Compliance Practice, Navigant; Treasurer, ETHICS; Former Global CCO, Siemens Healthcare, London, UK |

Gérald Hucky
Head Group Compliance, Sonova Group, Former Compliance Audit Manager, NameZimmer Biomet, Zürich, Switzerland |

Brett Hudson, MBIM
Head of Group I&C Group Integrity & Compliance, Novartis, Former Global Program Director - Integrity & Compliance, Sandoz, Basel, Switzerland
|

Arielle Hung
Compliance Counsel, Merck KGaA, Darmstadt, Germany |

Franziska Janorschke
Global Head Business Practices Office, Novartis International AG, Basel, Switzerland |

Carl Judge
Partner, Forensic & Integrity Services, EY, London, UK
|

Daniel T. Kessler, MA, JD
Founder & Principal, Givon Advisors; Former Chief Counsel, Int'l Trade Controls, Pfizer; Tel Aviv, Israel |

Timur Khasanov-Batirov, LLM
Compliance Officer, Russia, CIS & Romania, Dr. Reddy's Laboratories, Author, Compliance Man of Integrity Corp. and Integrity Corp. 50 Tips for Your Compliance, Program in the Post-Soviet States, Moscow, Russian Federation |

Maria Khoury, MBA, PharmD
Compliance Officer Levant, Roche, Chair, Lebanese Group of International Pharmaceutical Companies and Iraq LAWG (Local Area Working Group), Beirut, Lebanon
|

Anita Kyung-Hee Kim-Reinartz
Partner, Forensic & Integrity Services, EY, Düsseldorf, Germany |

Irith Kist, LLM, CIP/M
Functionaris Gegevensbescherming/ Data Protection Officer, The Netherlands Cancer Institute, Amsterdam, The Netherlands |

Katharina Kneisel, MBA
Senior Manager Corporate Compliance, Fresenius SE & Co. KGaA, Bad Homburg vor der H�he, Hessen, Germany
|

Tomasz Kruk
Head of Compliance, Vifor Pharma, Switzerland |

Chrystel Lanxade
Regional Compliance Officer, Europe Middle East Africa, Alcon; Former Head of Legal & Compliance Alcon France, Legal & Compliance Head Novartis Consumer Health France, Legal Counsel, Leem; Lawyer at the Paris bar, DEA, DESS; Cointrin, Canton of Geneva, Switzerland |

Aline Lautenberg
General Counsel and Director Legal & Compliance, MedTech Europe, Brussels, Belgium
|

Edoardo Lazzarini, PhD
Healthcare Compliance Consultant, Former EuCan Ethics & Compliance Initiatives Director, Takeda Pharmaceuticals, Former Compliance Officer EMEA (Europe, Middle East & Africa), Zimmer Biomet, Milan, Italy |

Martina Leposa
HealthCare Compliance Officer, Janssen-Cilag Ltd. (a Johnson & Johnson company), Chair, Compliance Committee, Association of Innovative Pharmaceutical Manufacturers, Budapest, Hungary |

Carlos Romero Lopez, LLB, MA
Head of Compliance Healthcare South Europe, Merck Group, Madrid, Spain
|

Michael K. Loucks, JD
Partner, Skadden Arps LLP, Former Acting United States Attorney, District of Massachusetts, US Department of Justice, Washington, DC, USA |

Paul J. Melling, JD
Founding Partner, Baker & McKenzie -- CIS, Limited, Moscow, Russia |

Laetitia Ducroquet Minel
Senior Director, Ethics & Compliance, Ipsen, Boulogne-Billancourt, France
|

Laura Nassar, PharmD
Head of Compliance Middle East, Roche Pharmaceuticals, Chair, Compliance Committee, Pharmaceutical Research, and Manufacturers Association, Gulf (PhRMAG), Beirut, Lebanon |

Michael O'Connor, MS
Vice President of Compliance Technology, Porzio Life Sciences, LLC, Former Global Head Compliance and Ethics Operations, Alexion, Former Executive Director, Global Head, IS Business Consulting, Boehringer Ingelheim, New York, NY |

Nicola Orlandi
Head, Data Privacy Pharma, Global Privacy Office, Novartis International AG; Former Head of Legal and Compliance, Sandoz S.p.A., Basel, Switzerland
|

Minal Patel
Health Care Compliance Officer, Pharma and Consumer, Johnson & Johnson, Johannesburg Area, South Africa |

Marie-Claire Pickaert
Former Deputy Director General, Chief Financial Officer and Ambassador to the Medical Communities, European Federation of Pharmaceutical Industries and Associations (EFPIA), Brussels, Belgium |

Madina Plieva, PhD
Legal and Compliance Head, Russia, Bristol-Myers Squibb, Former Legal Director, Association of International Pharmaceutical Manufacturers (AIPM), Former Head of Legal Department, Kutafin Moscow State Law Academy, Moscow, Russia
|

Mario Prohasky
Global Compliance Consultant, IQVIA, London, UK |

Ariadna Quesada, MSJ
Law, Ethics and Compliance Officer, The Netherlands, AbbVie; Former Compliance Manager International, MicroPort Orthopedics Inc., Amsterdam, Netherlands |

Alexander Roussanov, LLM
Counsel, Life Sciences, Hogan Lovells, Former Legal Adviser, European Medicines Agency, Brussels, Belgium
|

Giuseppe Saporito, LLM
Global Legal & Regulatory Expert, IQVIA, Milan, Italy |

Antje Petersen Schmalnauer, JD, LLM
Group General Counsel, Sonova Group, Former Interim VP Legal and General Counsel, Kautex Textron, Former Global Privacy and Data Protection Officer, Biomet, Cologne, Germany |

Leonardo Silva, LLM
Global Compliance Officer, Head of Global Compliance, Ferring, Former Head of Compliance, Acino Group, Former Compliance Director, Emerging Markets, Takeda, St. Prex, Switzerland
|

Ruth Sturm
Head of Compliance EMEA, SIEMENS Healthineers, Erlangen, Germany |

Ioannis Talaslis
Compliance Excellence Manager, Merck Serono GmbH, Offenbach am Main, Germany |

Kathy Tench, MS
Director, Life Sciences Compliance, KPMG Switzerland, Former Senior Director of Compliance, Optimer Pharmaceuticals, Z�rich, Switzerland
|

Eirini Tsotra, MS, CIPP/E, CIPM, CISA
Director, Privacy & Data Protection, Ferring International Centre SA, St-Prex, Switzerland |

Tamara Tubin
International Compliance Director, Corporate Compliance, Wright Medical Group N.V., Vice Chair, Ethics & Compliance Committee, MedTech Europe, Member, Strategic Committee, ETHICS, Zürich, Switzerland |

Satnam Tumani, LLB
Partner, Kirkland & Ellis International LLC, Former Head of Bribery & Corruption, Serious Fraud Office, London, UK
|

Katharina Weghmann, MBA, EdD
Associate Partner, Forensic & Integrity Services, EY, Düsseldorf, Germany |

Mariusz Witalis
Partner, Forensic & Integrity Services, EY, Warsaw, Poland
|

Niels Wohlwend, Master of Law (lic iur), MAES
Senior Director Healthcare Compliance (HCC) Europe & Canada, Celgene, Member, WFPIA Ethics & Compliance Committee, Former Global Head of Compliance, ACINO, Former Head of Compliance APAC, LATAM and MEA, Mundipharma, Boudry, Canton of Neuch�tel, Switzerland |

Elisabethann Wright, LLB
Partner, Hogan Lovells, Former, Senior Legal Officer and Hearing Officer, EFTA Surveillance Authority, Brussels, Belgium
|
|
|
|
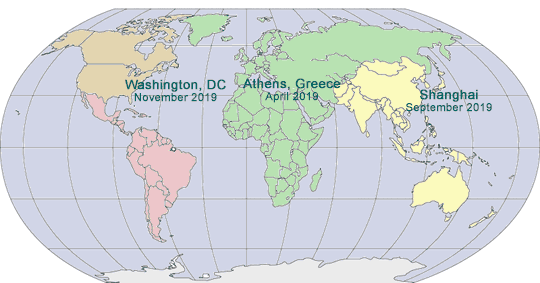
2019 GLOBAL PHARMA COMPLIANCE CONGRESSES
|
|
THIRTEENTH INTERNATIONAL PHARMACEUTICAL AND MEDICAL
DEVICE COMPLIANCE CONGRESS
|
Sponsored by International Society of Healthcare Compliance Professionals (ETHICS)
Media Partners: Policy & Medicine Compliance Update and Rx Data News
April 8 - 10, 2019
Grand Hyatt Athens
Athens, Greece
www.International PharmaCongress.com
|
|
NINTH ASIA PACIFIC
PHARMACEUTICAL AND MEDICAL
DEVICE COMPLIANCE CONGRESS
|
Sponsored by Asia Pacific Healthcare Industry Compliance Team
Cosponsored by International Society of Healthcare Ethics and Compliance Professionals (ETHICS)
Media Partners: Policy & Medicine Compliance Update
September, 2019
Shanghai, China
www.Asian PharmaCongress.com
|
|
TWENTIETH ANNUAL PHARMACEUTICAL AND
MEDICAL DEVICE COMPLIANCE CONGRESS
|
A Hybrid Conference and Internet Event
Sponsored by Pharmaceutical Compliance Forum
Media Partners: Harvard Health Policy Review, Health Affairs, Policy & Medicine Compliance Update and Rx Data News
November 6 - 8, 2019
Mandarin Oriental
Washington, DC
www.PharmaCongress.com
|
|
|
|
|
|
SPONSORED BY:
|

|
|
CO CHAIRS
|

Dante Beccaria
Senior Vice President, Global Compliance Officer, Head of Ethics & Business Integrity Department, Sanofi, Paris, France

Stephen Nguyen Duc
Vice-President, Global Head of Human Resources and Ethics & Compliance, Medday Pharmaceuticals; Board Member, Strategic Committee International Society of Healthcare, Ethics, and Compliance Professionals (ETHICS), Paris, France

Suzanne Durdevic, LLM
Associate General Counsel - International, Fortive/pending ASP acquisition; Member, Strategic Committee, International Society of Healthcare, Ethics and Compliance Professionals (ETHICS), Paris, France

Dominique Laymand, Esq.
Executive Vice President, Chief Ethics and Compliance Officer and CSR, Ipsen; Honorary President, International Society of Healthcare Ethics and Compliance Professionals (ETHICS), Paris, France

Roeland Van Aelst
Regional Vice President - HCCO MD&D EMEA & Canada, Office Health Care Compliance and Privacy, Johnson & Johnson, Chairman, International Society of Healthcare, Ethics and Compliance Professionals (ETHICS), Chairman, MedTech Compliance Network, Brussels, Belgium
|
|
GRANTORS
|
|
DIAMOND
|

|
|
SILVER
|


|
|
BRONZE
|








|
|
MEDIA PARTNER:
|


|
|
CONTINUING EDUCATION CREDITS:
|
Accounting Professionals: Approved for up to 18.5 NASBA CPE credits.
Attorneys: Approved for up to 14.00 Pennsylvania CLE Credits.
Compliance Professionals: The Compliance Certification Board (CCB)® has approved this event for up to 21.9 live CCB CEUs. Continuing Education Units are awarded based on individual attendance records. Granting of prior approval in no way constitutes endorsement by CCB of this event content or of the event sponsor.
Click here for more information.
|
|
STAY CONNECTED
|


Tweet using #PharmaCongress
|
|
THE CONGRESS IS
|

|
|
PAST GLOBAL PHARMA COMPLIANCE CONGRESSES
|

2007 - Brussels

2008 - Paris

2009 - Rome

2010 - Berlin

2011 - Istanbul

2011 - Singapore

2012 - Budapest

2012 - Shanghai

2012 - S�o Paulo

2013 - Madrid

2013 - Kuala Lumpur

2014 - Dubai

2014 - Mexico City

2014 - Shanghai

2015 - Manila

2016 - Warsaw

2017 - Lisbon

2018 - Vienna
|
|
ONSITE ATTENDANCE
|
Simply register, travel to the conference city and attend in person.
Pros: subject matter immersion; professional networking opportunities; faculty interaction

|
|
|
|
|
|
|
|