|
|
|
Register for 14th Int'l Pharma & Med Device Compliance Congress by 2/21 for Individual & Group Early Bird Rate
|
- Sponsored by ETHICS, the International Society of Healthcare Compliance Professionals
- Media Partners: Policy & Medicine Compliance Update and Rx Data News
- April 20 - 22, 2020
- Onsite at the Sheraton Munich Arabellapark Hotel
- Munich, Germany
- www.InternationalPharmaCongress.com
PRESS RELEASE
Phone: 800-503-8171
Email: registration@hcconferences.com
Website: www.InternationalPharmaCongress.com |
EARLY BIRD REGISTRATION
- SAVE UP TO €400 -
|
Register by Friday, February 21, 2020
save up to €400.
Click here to register.
|
AGENDA AND BROCHURE
NOW AVAILABLE
|
Click here to view the conference agenda.
Click here to view the conference brochure.
|
|
|
MUNICH, GERMANY -- PHARMA UPDATE NEWS SERVICE™ -- FEBRUARY 12, 2020: The Fourteenth International Pharmaceutical and Medical Device Ethics and Compliance Congress, www.InternationalPharmaCongress.com, will be held April 20 - 22, 2020 at the Sheraton Munich Arabellapark Hotel in Munich, Germany.
|
|
KEYNOTE SPEAKERS
|

Hani Abouhalka, MBA
Company Group Chairman, Medical Devices EMEA, Johnson & Johnson, New Brunswick, NJ, USA |

Gemma Aiolfi
Head of Compliance, Corporate Governance and Collective Action, Basel Institute on Governance; Former Advisor, Working Group on Bribery in International Business Transactions, Organisation for Economic Co-operation and Development (OECD); Basel, Switzerland |

Julie Bonhomme
EFPIA Legal & Compliance Director, European Federation of Pharmaceutical Industries and Associations (EFPIA), Brussels, Belgium
|

Robert I. Dodge, JD
Assistant Director, FCPA Unit, US Securities & Exchange Commission; Former Assistant U.S. Attorney, US Attorney's Office, Western District of Michigan; Former Assistant Section Chief, Environmental Defense Section, US Department of Justice; Washington, DC, USA |

Roxana Family, Thčse de Doctorat Droit
Vice-President, Chair in Law & Business Ethics and Program Director, Cergy Pontoise University, Cergy-Pontoise, France |

Susanne Friedrich, MA
Director, Group Governance, Human, Rights Division Global Policy, Governance, Alliance for Integrity (AfIn), Bonn, Germany
|

David Fuhr, JD
Assistant Chief in the FCPA Unit, US Department of Justice, Washington, DC, USA |

Iordanis Kerenidis, PhD
Director, Paris Centre for Quantum Computing (PCQC), CNRS Senior Researcher (DR2), Algorithms and Complexity Group, IRIF, University Paris Diderot, Paris, France |

Aline Lautenberg
General Counsel and Director, Legal & Compliance, MedTech Europe, Brussels, Belgium
|

Sofie Melis
Head of Ethics and Compliance, International Federation of Pharmaceutical Manufacturers and Associations (IFPMA), Geneva, Switzerland |

Hannes Oswald-Brugel, LLM, PhD
Global Head Pharma Healthcare Compliance, Roche, Vice President, Association of Voluntary Self-Regulation, for the Pharmaceutical Industry ("FSA"), Basel, Switzerland |

Christoph Thalheim
Vice CEO and Director External Affairs, European Multiple Sclerosis Platform (EMSP), Brussels, Belgium
|
|
|
|
FEATURING INVITATION-ONLY: CHIEF COMPLIANCE OFFICER ROUNDTABLE
|
(Special April 20 Morning Invitation-only, Closed Session)
SPONSORED BY

|
CO-SPONSORED BY


|
|
FEATURING
|

Sally Molloy, JD
Chief, Strategy, Policy & Training Unit, Fraud Section, Former Assistant U.S. Attorney, US Attorney's Office, Northern District of Georgia, US Department of Justice, Washington, DC
|
|
|
|
AGENDA-AT-A-GLANCE
|
|
Monday, April 20, 2020
|
- Morning Preconferences:
- Closed-Door, Invitation-Only Chief Compliance Officer Roundtable
- ETHICS Workshop on Practical Ethics & Compliance Case Studies
|
|
- Afternoon Opening Plenary Session:
- Congress Welcome, Vision and Overview
- Keynote Address: Ethics, Compliance and the C-Suite
- International Anti-Corruption Landscape
- Senior Ethics & Compliance Professionals Roundtable
|
|
|
Tuesday, April 21, 2020
|
- Morning Plenary Session:
- Embedding Ethics in a Company Governance Model
- Global Compliance Codes Update
|
- Minis Summits:
- I Compliance Considerations for Small to Mid-Sized Organizations
- II Annual Medical Device Compliance Roundtable
- III Interconnectivity of Auditing, Monitoring, and Investigations
- IV Annual Central and Eastern Europe (CEE) Compliance Update
- V Best Practices for Compliance Training
- VI How to Build and Manage Compliance Risk Assessments
- VII Handling Ethics & Compliance When Interacting with 3rd Parties
- VIII EU Whistleblower Directive
|
|
- Minis Summits, Continued:
- IX Performing a Fair Market Value (FMV) Analysis
- X Compliance Considerations for Gene Therapy& Rare Disease Products
- XI GDPR and the Tumultuous World of Data Privacy
- XII Patient Relationships, Including Patient Access and Support
- XIII Enhanced Compliance Monitoring
- XIV Building Effective and Right Sized Compliance Programs
- XV Role of Compliance Officers in Complex Global Organizations
|
- Afternoon Plenary Session:
- Value-Based Proposition
- Ethical Interactions between Health Care & Other Stakeholders
- Ethics and New Technologies
|
|
|
Wednesday, April 22, 2020
|
- Mini Summits:
- XVI Annual Middle East Africa (MEA) Compliance Update
- XVII The Role of the Industry re: Medical Education
- XVII The Practical Effect on Implications of Brexit
|
|
- Closing Plenary Session:
- Drivers for a Sustainable Ethical Culture
- Trust in Business
- The Role of Governance
- Co-chairs Closing Thoughts
|
|
|
FEATURED FACULTY
|

Pablo Rojas Abad, LLM
Manager, Legal and Compliance, MedTech Europe, Brussels, Belgium |

Bas Amesz
Partner, Vintura, Baarn, Utrecht, Netherlands |

Natalie Ammerlaan, LLM
Compliance Director International Operations, Novo Nordisk, Zürich, Switzerland
|

Michael Bartke, PhD
Member, Strategic Committee, ETHICS; Former Director Ethics & Compliance, Alexion; Former Director Compliance Management, Daiichi Sankyo Europe; Munich, Germany |

Dante Beccaria
Global Compliance Officer and Vice President, Sanofi; Former Vice President of Internal Audit, Sanofi; Former Co-chair, International Pharmaceutical and Medical Device Compliance Congress; Paris, France |

Enno Behrendt
Associate Director, Guidehouse; Former Head of Compliance for Diagnostic Imaging, Siemens Healthcare GmbH; Berlin, Germany
|

Sadek Beloucif, MD
University Professor, Hospital Practitioner and Head of Anesthesia-Resuscitation, University of Paris and University Hospital, Bobigny, Chair, National Union of Physicians, Surgeons, Specialists, and Biologists of Public Hospitals, Paris, France |

Duygu Beyazo
Senior Associate, NSN Law Firm; Former Compliance Manager (Secondment), Business Conduct, Gilead Sciences; Istanbul, Turkey |

Stefano Biondi, LLB, PhD
Group Data Protection Officer, Menarini Group, Florence, Italy
|

Eric Bolesh
Chief Operating Officer, Cutting Edge Information, Raleigh-Durham, NC, USA |

Kara Bonitatibus, JD
Head of Product, Lextegrity; Former Assistant General Counsel, Compliance Lead for Digital, Policy, and Patient & Health Impact, Pfizer, New York, NY |

Keith Burn
Global Investigations Director, Ipsen; Former Associate Investigator, Parliamentary and Health Service Ombudsman, NHS; Former Detective Constable, London Metropolitan Police; Amersham, Slough, UK
|

Parth Chanda, JD, MPA
Founder & CEO, Lextegrity; Former Global Anti-Corruption Lead and Chief Compliance Counsel, Oncology, Pfizer; New York, NY, USA |

Alice Choy, MS
Manager, Anti Bribery Anti-Corruption (ABAC), Compliance, Internal Audit, Risk Assessment, IQVIA; Former Lead Specialist, Transparency and Reporting, Johnson & Johnson; London, UK |

Thomas Costa, JD
Member, US Board of Directors, Sanofi; Former Vice President, US Compliance & Ethics, Bristol-Myers Squibb; Washington, DC, USA
|

Holger Diener
Managing Director, Association of Voluntary Self-Regulation for the Pharmaceutical Industry ("FSA"), Member, Ethics and Compliance Committee and Vice Chair, Code Committee, EFPIA, Member, IFPMA Code Compliance Network, Berlin, Germany |

Peter Dieners, Esq.
Partner and Head, Global Healthcare and Life Sciences Group, Clifford Chance, Co-chair, Legal Affairs Focus Group (LA FG), EUCOMED, Co-chair, Compliance Network (CN), EUCOMED, Düsseldorf, Germany |

Julien Durand, MBA, PhD
Vice President, Head of Transformation, Global Ethics and Compliance, Takeda, Chair, IFPMA Future Health Technologies & Bioethics Working Groups, Former Executive Director, Compliance and Ethics, Amgen, Former Chief Privacy Officer and Global Technology Compliance Head, AstraZeneca, Zürich, Switzerland
|

Sue Egan
Director and Principal Consultant, Sue Egan Associates; Former Vice President Compliance, AstraZeneca; Great Missenden, Buckinghamshire, UK |
Liisa Eisenlohr, PhD, MBA, CCRA, BCMAS
Associate Director, Guidehouse; Former Senior Director, Global Medical Information, Clovis Oncology, London, UK (Discussion Coordinator) |

Karen Eryou
Head, Ethics & Compliance Programs, Global, UCB Pharma; Former Co-chair, Asia Pacific Pharma Compliance Congress; Brussels, Belgium
|

J. Mark Farrar, MSJ, CPA, CFE, CFF
Partner, Life Sciences Governance, Risk Management and Compliance, Guidehouse, Atlanta, GA |

Alex Fell
Head Ethics and Compliance International; Amicus Therapeutics; Former VP Global Ethics & Compliance, Head of Strategy, Planning and Operations, GSK; London, UK |

George Fife
Partner, Forensic & Integrity Services, EY; Former Executive Director, Compliance & Ethics, Bristol-Myers Squibb; Paris, France
|

Indrani M. Lall Franchini, JD
Executive Vice President & Chief Compliance Officer, Alexion Pharmaceuticals, Inc.; Former Assistant General Counsel, Pfizer; New York, NY, USA |

Caroline Franco, MBA
Ethics and Compliance Manager, Boehringer Ingelheim, Amsterdam, Netherlands |

Abhiroop Gandhi
Trust and Compliance Officer, Verily Life Sciences (an Alphabet company); Former Vice President, Corporate Compliance, Mallinckrodt; San Francisco, CA, USA
|

Klaus Geldsetzer, MBA
Chief Compliance Officer EMEA, Santenb, Chairman, Eye-Care Industries European Economic Interest Grouping; Former Head of R&D Office and Global Project Leader, Allergan Ltd; Munich, Germany |

Gary F. Giampetruzzi, Esq.
Partner, Paul Hastings; Former Vice President and Assistant General Counsel, Head of Government Investigations, Pfizer Inc.; New York, NY, USA |

Laura Coll Gil, LLM
Ethics and Compliance Officer, Boehringer Spain, Barcelona, Catalonia, Spain
|

Betania Glorio, LLM
Global Compliance Officer, Healthcare, Merck KGaA; Former Senior Director, Legal and Compliance EMEA, Horizon Pharma plc and Raptor Pharmaceuticals; Darmstadt, Germany |

Tom Gregory, MBA
Partner, Forensic & Integrity Services, EY, Atlanta, GA, USA |

Ulf H. Grundmann
Partner, FDA and Life Sciences, King & Spalding LLP, Lecturer, Frankfurt School of Finance and Management, Frankfurt am Main, Germany
|

Joseph W. Henein, PharmD
President and Chief Executive Officer, NewBridge Pharmaceuticals, Dubai, UAE |

Casey J. Horton, CFE
Director, Life Sciences, Governance, Risk and Compliance, Guidehouse, Chicago, IL |

Bella Hovhannisyan
Business Integrity and Compliance Lead, Intercontinental Commercial Operations, CSL Behring, Bern, Switzerland
|

Mirgen Jaku, MA
Lead of Market Readiness, Digital Surgery, EMEA, Johnson & Johnson, Hamburg, Germany |

Franziska Janorschke
Global Head, Business Practices Office, Novartis International AG, Basel, Switzerland |

Els Janssens, LLM
Associate, Baker & McKenzie; Former Legal Adviser, European Medicines Agency; Former Senior Legal Counsel, Johnson & Johnson; Brussels, Belgium
|

Anita Kyung-Hee Kim-Reinartz
Partner, Forensic & Integrity Services, EY, Düsseldorf, Germany |

Elisabeth Kohoutek
Senior Associate, FDA and Life Sciences, King & Spalding LLP, Frankfurt am Main, Germany |

Adem Koyuncu, MD, PhD (Law)
Partner and Chair, Food, Drug & Device Practice Group, Covington, Brussels, Belgium
|

Tomasz Kruk, LLM, MBA
Head of Compliance, Vifor Pharma; Former Director International Compliance, Mallinckrodt; Former Director Global Ethics & Compliance, Actavis; Zürich, Switzerland |

Edoardo Lazzarini, PhD
Healthcare Compliance Consultant; Former EuCan Ethics and Compliance Initiatives Director, Takeda Pharmaceuticals; Former Compliance Officer EMEA (Europe, Middle East & Africa), Zimmer Biomet; Milan, Italy |

Lei Li, LLM
Managing Partner, Beijing and Shanghai Offices, Sidley Austin; Former Third Secretary, Ministry of Commerce, People's Republic of China, Beijing, China; Beijing, China
|

Bruno Lichtinger
Managing Director, INTERPLAN Congress, Meeting & Event Management AG, Munich, Germany |

Michael K. Loucks, JD
Partner, Skadden Arps LLP; Former Acting United States Attorney, District of Massachusetts, United States, Department of Justice; Washington, DC, USA |

Marcel Maderitsch, LLM
Senior Compliance Officer, Vifor Pharma; Former Compliance Manager EMEA, Zimmer Biomet; Zurich, Switzerland
|

Rosa Magistri, Esq., CIPP/E
Head of Legal and Compliance Europe, SeaGen International GmbH; Former Regional Director Office of Ethics and Compliance, Western Europe & Canada, Region South & Israel, AbbVie; Former Associate Compliance Director, Shire; Zug, Switzerland |

Anthony McQuillan, LLB
Vice President Legal & Compliance EMEA, Medtronic International; Former Senior Attorney EMEA, Hewlett-Packard; London, UK |

Paul J. Melling, JD
Founding Partner, Baker & McKenzie – CIS, Limited, Moscow, Russia
|

Laetitia Ducroquet Minel
Senior Director, Business Ethics, Ipsen, Paris, France |

Véronique Monjardet, MS, DEA, PhD
Sales Director, IQVIA, Paris, France |

Laura Nassar, PharmD
Vice President, Head of Ethics & Business Integrity, AEME Region, Sanofi, Chair, Compliance Committee, Pharmaceutical Research, and Manufacturers Association, Gulf (PhRMAG); Former Head of Compliance Middle East, Roche Pharmaceuticals; Beirut, Lebanon
|

Lauri G. Opar
Director, Risk Analytics, Global Ethics & Compliance, Glaxosmithkline, Brentford, Middlesex, UK |

Pascale Paimbault
CEO, Consulting Alley, Strategic Committee Member, ETHICS; Former Executive Director, EMEA Compliance Astellas Pharma Inc.; Former Compliance Officer, France -Benelux, Biogen; Former Chief Compliance Officer EMEA, Wright Medical Inc.; Former Senior Director, EMEA, Bristol-Myers Squibb; Paris, France |

Giota Papamarkou
Vice President, Business Ethics Global, France, Ipsen; Former EMEA Compliance and Ethics Manager, Bristol-Myers Squibb, Paris, France
|

Piergiorgio Pepe, MA
EU Law, President, Quantum Ethics, Ethics and Compliance Lecturer, SciencesPo; Former Compliance Director Western Europe & Canada, AbbVie; Former Director, Compliance & Ethics EMEA, Bristol-Myers Squibb; Paris, France |

Oscar Perdomo
Director, Pharmaceutical & Life Sciences Advisory Services, PwC Switzerland, Zurich, Switzerland |

Mario Prohasky
Global Compliance Consultant, IQVIA, London, UK
|

Ariadna Quesada, MSJ
Law, Ethics and Compliance Officer, The Netherlands, AbbVie; Former Compliance Manager International, MicroPort Orthopedics; Amsterdam, Netherlands |

John Seungjoo Rah, JD
Partner, DLA Piper, Washington, DC |

Nadčge Rochel
Global Compliance Manager, Hollister Incorporated, Milan, Italy
|

Christian-Claus Roth
Head Scientific Engagement Governance, Novartis, Basel, Switzerland |

Matteo Santi
Group Compliance Officer, Helsinn Healthcare, Ticino, Switzerland |

Giuseppe Saporito, LLM
Global Legal & Regulatory Expert, IQVIA, Milan, Italy
|

Pascale Schmidt
Chief Compliance Officer, F. Hoffmann-La Roche Ltd, Basel, Switzerland |

Piyush Sharma, JD
Senior Vice President, Deputy Chief Compliance Officer, International Compliance, Alexion Pharmaceuticals, New Haven, CT |

Leonardo Silva, LLM
Vice President, Global Chief Compliance & Privacy Officer, Ferring; Former Head of Compliance, Acino Group; Former Compliance Director, Emerging Markets, Takeda; Lausanne, Switzerland
|

Robert Stephenson
Senior Manager, PwC UK, London, UK |

Madina Torchinova
Vice President, Deputy Head Global Compliance, MorphoSys; Former Regional Head of Compliance, Central & Eastern Europe, Middle East & Africa, and Global OTC, Sandoz; Munich, Germany |

Tamara Tubin
International Compliance Director, Corporate Compliance, Wright Medical Group N.V., Vice Chair, Ethics & Compliance Committee, MedTech Europe, Member, Strategic Committee, ETHICS, Zürich, Switzerland
|

Cláudia Vaz
Global Value Based HealthCare Consultant, Roche, Value-Based Health Care Consultant, International Consortium for Health, Outcomes Measurement (ICHOM), Basel, Switzerland |

Samar Wakim, PharmD
Ethics, Risk & Compliance Head MEA, Oncology, Novartis, Dubai, UAE |

Vanessa Westphal, JD, LLM
Head of Compliance Programs & Support, Merck KGaA, Frankfurt am Main Area, Germany
|

Christopher L. White, JD
Chief Operating Officer and General Counsel, Advanced Medical Technology Association (AdvaMed), Washington, DC, USA |

Mariusz Witalis
Partner, Forensic & Integrity Services, EY, Warsaw, Poland
|

Niels Wohlwend, LLM
Senior Director Healthcare Compliance, Europe & Canada, Celgene, Member, Ethics & Compliance Committee, EFPIA; Former Global Head of Compliance, ACINO; Boudry, Switzerland |

Elisabethann Wright, LLB
Partner, Hogan Lovells; Former Senior Legal Officer and Hearing Officer, EFTA Surveillance Authority; Brussels, Belgium
|
|
|
|
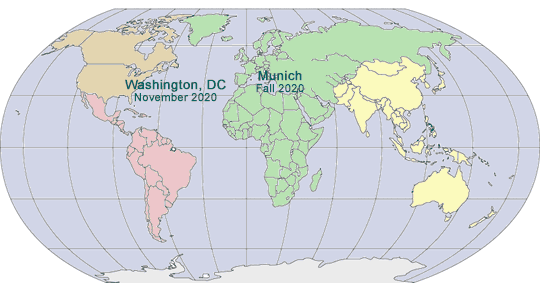
2020 GLOBAL PHARMA COMPLIANCE CONGRESSES
|
|
FOURTEENTH INTERNATIONAL PHARMACEUTICAL AND MEDICAL DEVICE COMPLIANCE CONGRESS
|
Sponsored by International Society of Healthcare Compliance Professionals (ETHICS)
April 20 - 22, 2020
Sheraton Munich Arabellapark Hotel
Munich, Germany
www.International PharmaCongress.com
|
|
TWENTY-FIRST ANNUAL PHARMACEUTICAL AND MEDICAL DEVICE COMPLIANCE CONGRESS
|
A Hybrid Conference and Internet Event
Sponsored by Pharmaceutical Compliance Forum
November 4 - 6, 2020
Mandarin Oriental
Washington, DC
www.PharmaCongress.com
|
|
|
|
|
|
SPONSORED BY:
|

|
|
CO CHAIRS
|

Anne-Sophie Bricca, MA, DEA International Law, DES European law
Deputy General Counsel & Senior Director Legal Affairs & Compliance, Terumo BCT, Chair, Ethics and Compliance Group and seats at the Code Committee, MedTech Europe, Brussels Area, Belgium

Stephen Nguyen Duc
Vice-President, Global Head of Human Resources and Ethics & Compliance, Medday Pharmaceuticals; Board Member, Strategic Committee International Society of Healthcare Ethics and Compliance Professionals (ETHICS), Paris, France

Dominique Laymand, Esq.
Executive Vice President, Chief Ethics and Compliance Officer, Ipsen; Honorary President, International Society of Healthcare Ethics and Compliance Professionals (ETHICS), Paris, France

Roeland Van Aelst
Regional Vice President -- HCCO MD&D EMEA & Canada, Office Health Care Compliance and Privacy, Johnson & Johnson, Chairman, International Society of Healthcare, Ethics and Compliance Professionals (ETHICS), Chairman, MedTech Compliance Network, Brussels, Belgium
|
|
GRANTORS
|
|
SILVER
|




|
|
BRONZE
|










|
|
MEDIA PARTNER:
|

|
|
CONTINUING EDUCATION CREDITS
|
Accounting Professionals: Approved for up to 19 NASBA CPE credits.
Attorneys: Pending approval for PA MCLE credits.
Compliance Professionals: This education activity has been submitted to the Compliance Certification Board (CCB)® and is currently pending their review for approval of CCB CEUs.
Click here for more information.
|
|
STAY CONNECTED
|


Tweet using #PharmaCongress
|
|
ONSITE ATTENDANCE
|
Simply register, travel to the conference city and attend in person.
Pros: subject matter immersion; professional networking opportunities; faculty interaction

|
|
INTERNATIONAL PHARMA CONGRESS IS
|

|
|
|
|
PAST GLOBAL PHARMA COMPLIANCE CONGRESSES
|

2007 - Brussels

2008 - Paris

2009 - Rome

2010 - Berlin

2011 - Istanbul

2011 - Singapore

2012 - Budapest

2012 - Shanghai

2012 - Săo Paulo

2013 - Madrid

2013 - Kuala Lumpur

2014 - Dubai

2014 - Mexico City

2014 - Shanghai

2015 - Manila

2016 - Warsaw

2017 - Lisbon

2018 - Vienna

2019 - Athens
|
|
|
|
|